Alnylam Pharmaceuticals


ABOUT ALNYLAM
Our Science Is Changing the Way Medicine Treats DiseaseTM
Alnylam has led the translation of RNAi (RNA interference) from Nobel Prize-winning discovery into an innovative, entirely new class of medicines. Founded in 2002 by a team of distinguished life sciences leaders, Alnylam’s vision is to harness the potential of RNAi therapeutics to transform the lives of people living with diseases for which there are limited or inadequate treatment options. Our pioneering work has delivered the world’s first and only approved RNAi therapeutics—ONPATTRO® (patisiran) in 2018, GIVLAARI® (givosiran) in 2019, OXLUMO® (lumasiran) in 2020, and AMVUTTRA® (vutrisiran) in 2022. We are advancing a deep pipeline of innovative RNAi-based medicines in four therapeutic areas: genetic medicines, cardio-metabolic diseases, infectious diseases, and central nervous system (CNS) and ocular diseases.

What’s in a Name?
AL-NY-LAM. Our name may not be the easiest to pronounce, but once you learn it, you’ll never forget it. Alnylam is derived from “Alnilam,” the bright center star in the constellation Orion’s belt, which has been used by navigators for thousands of years and symbolizes our passion for discovery.
RNAi Therapeutics—An Innovative New Class of Medicines
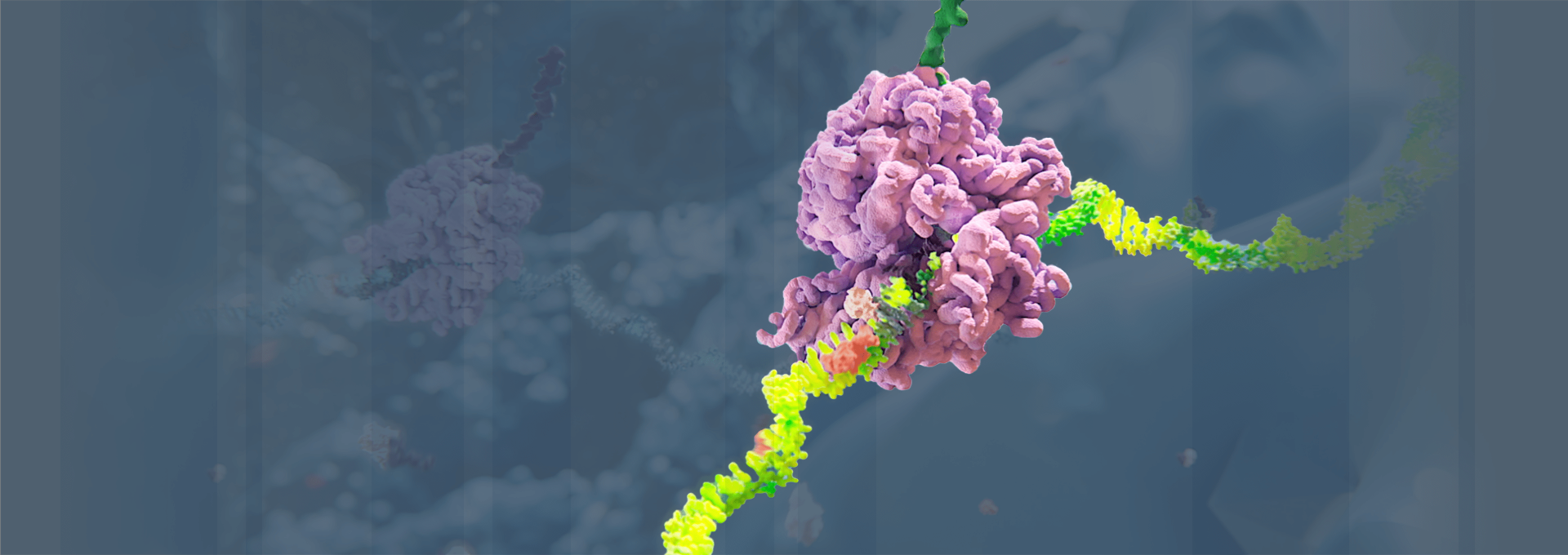
Medicines based on RNAi work by “silencing” or disabling the production (“expression”) of the genes that cause specific diseases. In doing so, RNAi therapeutics work “upstream” of most other classes of medicines, such as small molecules and monoclonal antibodies, by targeting the “root” genetic cause of a disease rather than its symptoms. To learn more about our science, click here.
RNAi is the core discovery that forms the therapies Alnylam is developing. It is recognized as a major scientific breakthrough—but how does it work, exactly?
Vision, Mission, and Values
Our Vision - Harnessing a revolution in biology for human health®.
Our Mission - Build a top-tier, global, independent biopharmaceutical company founded on RNAi.
Our Core Values - Commitment to People, Innovation & Discovery, Sense of Urgency, Open Culture, and Passion for Excellence.
CAREERS AT ALNYLAM
Join Our Team and Grow With Us
At Alnylam, we are driven by our mission to translate the Nobel Prize-winning discovery of RNA interference (RNAi) into an innovative, new class of medicines. We’re motivated in this endeavor by the bravery and perseverance of people with unmet medical needs who live with rare and genetic diseases. They are the focus of our efforts, and we exist as a company because of them. That knowledge and commitment to patients is present in everything we do, every day.

At The Forefront of What’s Next
For Patients
Our goal, which is well underway, is to become a world-class biopharmaceutical company that can independently develop and deliver innovative medicines based on RNAi to patients worldwide. We have a deep pipeline of early to late stage investigational therapies, and 2018 marked the approval of our first medicine—the world’s first RNAi therapeutic. Even though we’ve been around since 2002, we’re just getting started!
For Employees
We are growing quickly and hiring across North America, Europe, Asia, and Latin America for a wide variety of roles. We’re seeking smart, passionate, “change the world” kind of people who are ready to say, “challenge accepted.” Our 1,500 (and counting!) employees are building a diverse, equitable, inclusive, and high-performing global organization that celebrates team achievement and recognizes individual contributions.
We’re at the forefront of what’s next for patients, and perhaps your career also.
Search among Alnylam Pharmaceuticals jobs
| Jobs: 1 - 10 of 112 |

Business Account Executive - Rare Disease (Miami)
Cambridge, Massachusetts
Overview We are looking for an experienced and talented Business Account Executive (BAE) to commercially represent GIVLAARI and OXLUMO in the United States and have a significant impact on the lives of AHP patients. AHP's represent a sub-gr...
1h
| Job Type | Full Time |

Principal Data Engineer, Marketing
Cambridge, Massachusetts
Overview AlnylamPharmaceuticalsisbuildingadata-drivendigitalmarketingcapabilitythatintegratestechnologyandcommercialoperationstodeliverpersonalizedexperiencesforhealthcareprofessionalsandpatientsacrossourrarediseaseportfolio.TheAssociateDir...
1h
| Job Type | Full Time |

Business Account Executive-Rare Disease (Dallas)
Cambridge, Massachusetts
Overview We are looking for an experienced and talented Business Account Executive (BAE) to commercially represent GIVLAARIand OXLUMO in the United States and have a significant impact on the lives of AHP patients. AHP's represent a sub-gro...
1h
| Job Type | Full Time |

Business Account Executive (Chicago)
Cambridge, Massachusetts
Overview We are looking for an experienced and talented Business Account Executive (BAE) to commercially represent GIVLAARIand OXLUMO in the United States and have a significant impact on the lives of AHP patients. AHP's represent a sub-gro...
1h
| Job Type | Full Time |

Cambridge, Massachusetts
Overview We are seeking an Accounting Manager to play a key role in Alnylam's core accounting operations, including monthly close, audits, systems, and application of accounting policies in accordance with U.S. GAAP. This handson role requi...
1h
| Job Type | Full Time |

Associate Director, Data Stewardship
Cambridge, Massachusetts
Overview Join the Alnylam Commercial Data and Analytics team and help build a product-driven engine that turns data-driven insights into deployed decisions with measurable business impact. As part of Customer Experience and Innovation, you ...
1h
| Job Type | Full Time |

Scientist, In Vivo Pharmacology
Cambridge, Massachusetts
Overview We are seeking a highly motivated Scientist, In Vivo Pharmacology to support the development of innovative therapeutics for autoimmune diseases and oncology. The ideal candidate will have strong hands-on experience in preclinical i...
1h
| Job Type | Full Time |

Associate Director, Medical Science Liaison - Rocky Mountains
Cambridge, Massachusetts
Overview: We are seeking an experienced Medical Science Liaison (MSL) to join the TTR team and support Alnylam's commitment to advancing the science and care of patients with transthyretin (TTR) amyloidosis. Patients often experience prolon...
1h
| Job Type | Full Time |

Business Account Executive, TTR - Peoria, IL
Cambridge, Massachusetts
Overview We are looking for experienced Business Account Executives (BAEs) with specialty sales experience to promote AMVUTTRA. Patients with TTR amyloidosis often spend years undiagnosed and misdiagnosed. This is an amazing opportunity to ...
1h
| Job Type | Full Time |

Director, Corporate Social Responsibility
Cambridge, Massachusetts
Overview The Director of Corporate Responsibility will lead key components of Alnylam's corporate responsibility strategy, including philanthropic initiatives, employee engagement, and community partnerships. This role blends strategy, exec...
1h
| Job Type | Full Time |

